AGING DEFINED

Overall, the scientific definition of aging continues to evolve as researchers learn more about the underlying biological mechanisms involved. However, the general consensus is that aging is a complex and multi-factorial process that results in the decline of physiological functions and increased vulnerability to age-related diseases (Complete list at bottom of this page), that eventually cause death.
At the cellular level, aging is characterized by a decline in cellular function and an increase in cellular damage, such as DNA damage, oxidative stress, and inflammation. This leads to a decline in the ability of cells to repair themselves and maintain proper function, ultimately contributing to the aging of tissues and organs.
At the molecular level, aging is thought to involve changes in gene expression and the regulation of cellular processes, such as autophagy, which is the process by which cells degrade and recycle cellular components.
Aging is primarily controlled by two intersecting and interconnected systems, each directing the other through a feedback loop (See Illustration Below).
Chronokines represent all the age-communicating fractions of the proteome, which is an umbrella term for all proteins contained within plasma. As a subset of the proteome, Chronokins are a combination of two terms: "chronos," indicating time, and "kinesis," indicating movement. Cytokines, a broad category of small peptides and proteins ranging from 5 to 25 kDa, play an important roles in cell signaling and can act as immunomodulating agents in autocrine, paracrine, and endocrine signaling. These include chemokines, interferons, interleukins, lymphokines, and tumour necrosis factors. Chronokines communicate and control incremental and progressive network shifts, producing cellular and systemic changes that both reflect and dictate the biological age of organisms.
Epigenetic changes, as evidenced by methylation additions to CpG-DNA motifs, along with histone changes, progressively obstruct the accessibility of crucial genetic information. This leads to the progression of aging and the promotion of various diseases and sets the bench-mark for the organisms biological age.
Multiple experiments have clearly demonstrated that the aging phenotype in cells can be reversed by circulating factors (chronokines) in the blood. Emerging studies are also demonstrating that a process called Reprogramming Induced Rejuvenation (RIR) is capable of resetting / reprogramming the epigenetic age-set-point of any animal. This is being accomplished with transcription factors and small molecules.
Evidence of a two way communication connection between these systems comes from small animal research into reprogramming induced rejuvenation(RIR). RIR induces partial reprogramming, but at different rates and efficiencies. Regardless of the age-set-point initially achieved by each cell type, after a short period of time the entire organisms age gets normalized across the systemic spectrum of cell and organ types. This indicates that a factor is communicating and normalizing the systemic system to a consensus age. Harold Katcher’s plasma fractions also provides conformation of this effect, but from the other side of this two factor equation; chronokines.
This new definition provides two primary interventional points to reset the biological age: 1) Selectively removing the accumulating aging-related epigenetic marks, referred to as reprogramming; and/or 2) reintroducing a younger set of Chronokines to redirect the systemic cell state to a younger set-point. As the graphic above demonstrates, each is capable of directing/controlling the other.
Having an accurate definition of aging is critically important. If you can’t effectively define a problem, you have virtually no chance of sucessfully addressing it. The focus and rational of this entire site is defined by the definition of aging provided above.
Our definition focuses on a primary feedback loop that may or may not be the final product of a long cascade of biological signaling events. As new insights into the root cause of aging emerge, the definition above will be refined. Josh Mitteldorf has identified potential age-controlling processes that each have independent influences on cellular and systemic health. Josh’s blog, is one of the best sources of news and information on the emerging science of aging.
An epigenetic clock alters gene expression over time in directions that give rise to self-destruction.
A telomere clock, counting cell divisions on a flexible schedule, eventually produces cells with short telomeres that poison us.
The thymus, a crucial training ground for our white blood cells, shrinks over a lifetime.
A neuroendocrine clock in the hypothalamus
We addrerss age regression by transforming the currently available research from multiple lab groups, biopharmaceutical companies and individual researchers, into understandable and actionable interventions, easily implemented by anyone. To quote one prominent aging researcher; “What works, what’s real and what doesn’t.” To address aging, you first must appreciate the most recent discoveries researchers have provided, describing and defining what the biological process we call aging, actually consists of. To that end we provide above, our own amalgamation of the current definition(s) of aging, guided by the most current published insights. Of course there are other definitions held by credible scientist in the anti-aging field.
There is a critical point in the definition provided above that can be easily missed. Read the bare bones definition again. “Aging is controlled by age-determining signaling molecules regulating the Biological Age-Set-Point.” If signaling molecules (chronokines) are directing all of the cells of the body as to what their assigned age should be, then those signals can be intercepted, blocked and/or replaced with “younger,” biological age-set-point signals. This description is believed by many to be a primary or first order directive of the aging paradigm. That process is depicted in the schematic above.
In a 2005 Letter to Nature, the Conboy’s research lab demonstrated this principle in mice. Our experiments suggest that there are systemic factors that can modulate the molecular signalling pathways critical to the activation of tissue-specific progenitor cells, and that the systemic environment of a young animal is one that promotes successful regeneration, whereas that of an older animal either fails to promote or actively inhibits successful tissue regeneration. It will be of great interest to identify the factors that have such a critical influence on tissue-specific progenitor cells. Our studies also demonstrate that the decline of tissue regenerative potential with age can be reversed through the modulation of systemic factors, suggesting that tissue specific stem and progenitor cells retain much of their intrinsic proliferative potential even when old, but that age-related changes in the systemic environment and niche in which progenitor cells reside preclude full activation of these cells for productive tissue regeneration. [12]
⫸ The blood delivers nutrients, oxygen, hormones, and growth factors to all organs and tissues. Moreover, it also transmits orders to the entire body and controls the immune system [52]. An age-related change in the profiles of bloodcirculation factors reflects the processes and mechanisms of aging. The medical implementation of these factors into the bloodstream allows conveying signals to the internal body systems that regulate and monitor aging [27]. ⫷[9]
These processes are staged, that is non-linear. [10]
⫸ Harold Katcher wrote in 2013: if a cell is placed in a “young environment” or an “old environment” that cell will assume the age phenotype appropriate to its environment. In agreement with the effects of the systemic environment on aging, aged stem cells were rejuvenated by young plasma in vitro, and young stem cells were “aged” by in vitro exposure to plasma from old animals [32]. ⫷ [11] Harold Katcher’s most recent advances in the field of age regression can be reviewed on this page.
This observation makes the regressive treatment directive simple and achievable. Block the increasing, negative signals advancing the age-set-point and augment the decreasing, positive signals regressing the age-set-point backwards.
It should be pointed out at this point that we are not in any way ignoring the factors in control of this process. Increases in methylation “marks,” are directly associated with the advancement of aging. [13] Genetic control of the signaling milieu is not in conflict with the interventional goals or goal post described here. See: Epigenetic Reprogramming
There are effective age-regressive therapies that appear to contradict this definition. Gregory Fahy’s thymus regeneration protocol incorporating Human Growth Hormone (HGH), metformin, and DHEA to re-constitute the thymic microenvironment would be one example. But after further examination this also clearly falls within the definition provided above. The thymic involution/atrophy is the direct result of proteomic signaling. This decline in thymus function, in all probability, represents the first biological and clinical demarcation point where aging begins. That would put the starting gun of this deleterious pathway at around 15 years of age.
Currently multiple research groups are attempting to determine if any one, or any subset of the identified chronokines are deterministic for controlling the biological age set pont of an individual. It now appears that a subset of these chronokines hold the possibility of being first order age-determents.
The viability of each approach can now be easily confirmed utilizing epigenetic clocks. [1,2] Clinical observations, normal blood cell and chemistry panels can also provide valuable feedback and validation on the effectiveness of your own current interventional strategy. [3,4] These effective and highly correlated validating processes are confirming both that the definition of aging provided above is accurate, and that the targets identified within the articles and the articles referenced, are points where active interventions are addressable by a targeted armentarium of multiple modalities we collectively refer to as AIR. These Available Interventional Resources (AIR) include: Amino Acids, Hormones, Vitamins and Nutritional Supplements, Metals/Minerals, Pre and Probiotics, and Small Molecules/Drugs. That list would not be complete without the addition of diet, exercise, sleep and psychological support. The vast majority of the required targeted agents identified herein are readily available.
Changing the controlling directive of the plasma fraction proteins in the peripheral blood stream, signaling milieu, to a younger age-set-point is now a realistic and achievable goal.
Over the next few years expect an avalanche of new treatments targeting these pathways. Many of them will initially seem to be disease specific targets because the FDA is still developing a clear pathway to develop drugs that are specifically designed and designated to regress aging. At least for the short term, this requires developers to identify the most likely disease process that will be beneficially altered by these new modalities. If these interventions are targeting a disease associated with aging, utilizing the chronokines identified within this site, it will be easily understood by you, that the mechanism is regressing the BASP to ameliorate that one specific and targeted disease indication. It will also be understood that this new drug application will have broader anti-aging or BASP regressing capabilities. Until these drugs become readily available, AIR interventions are powerful and effective alternatives.
•••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••••
Aging is now emerging as a biologically segmented, incremental staged process that is harmonized, coordinated and synchronized at each step by intracellular and extracellular molecular signaling pathways, driving complex, multifaceted, positive/negative-feedback loops. [5]
All of these networks are entangled, even if they seem completely independent of each other. These networks control all physiological age-associated changes in all mammalian organisms. “These changes include proteomic, transcriptional and epigenetic modifications; changes in energy production within mitochondria; and alterations in the molecular biology or mechanistic interactions of cells, their nuclei, and their surrounding extracellular matrix. In addition, aging influences the ability of cells to sense changes in extracellular-matrix compliance (mechanosensation) and to transduce these changes into biochemical signals (mechanotransduction).”[6] It has now been definitively proven that the signaling pathways directing the aging pathways, (primary blood circulating factors), in all mammalian organisms can be redirected to regress the age state and amarillite the degenerative processes associated with aging, allowing for extension of both health and life spans.[7]
A principal controlling component of this network consist of signal molecules generated by intra and extra cellular factors transported to all cells by the plasma component of blood. This age defining system includes complex signal response loops, driven by multiple feedback signals and crosstalk between exogenous, molecular and genetic pathways. It is now possible to strategically amplify (up-regulate) or block (down-regulate) these signals redirecting the accretion aging paradigm. This enables us to utilize the same molecular and genetic pathway targets to mimic the cellular signals normally directing and driving the aging process. Biosimilar signaling agents can be introduced in the form of [a] small molecules or drugs, [b] biological agents, [c] nutritional foods and [d] supplements or nutraceuticals, [[8] to accomplish this goal.
Noise is also a contributing factor. Aging has multiple pathways that contribute to the network of signals controlling your BASP. The viability and health of cells diminishes during each cellular replication cycle. These cellular replications are necessary to replenish all tissues and organs in our bodies. During each replication cycle, genetic information can be lost through a variety of mechanisms. Degradation of the genetic code also degrades the cells viability impacting its health, functionality and biological age. This process can be referred to as Reproductive Noise Damage (RND). An insightful doctoral dissertation; “Reversal of aging via in vivo epigenetic reprogramming,” by Yuancheng Lu. provides an excellent and detailed description of this process.
The network of differentially expressed genes (DEGs) as well as the progressive silencing of genes, are also a driving confluence that both receives signals and direct protein and chronokine instructions. DNA can be directly methylated through the covalent linkage of a methyl group to the fifth position of the cytosine ring to generate 5-methylcytosine (5mC). This modification is mainly present in DNA regions rich in cytosine-phospho-guanine (CpG) dinucleotides. While there is extensive evidence that DNA methylation at promoter regions is associated with gene silencing, the decipherment of the function of gene body methylation is still ongoing [15].
The selective pressures influencing our understanding of aging in the perspective of this stochastic storm of crosstalk between exogenous and endogenous signals, molecular pathways and gene signaling has not been completely elucidated, but candidates, those with clearly evident influence on the aging process and thus the most obvious and efficacious targets of immediate intervention have been identified enabling a new level of control of our biological age.
The information is available now to impact the aging process in a nominal way. As interventional information emerges from the ongoing research the ability to intervene in more dramatic way will become available in a time frame that will shock everyone.
REFERENCES:
[1] [2020] DNA Methylation Biomarkers in Aging and Age-Related Diseases
[2] [2019] DNA methylation GrimAge strongly predicts lifespan and healthspan
[3] Insilico Medicine Aging.AI
[4] Ageless Rx Bloodtest
[5] [2020] Cell non-autonomous regulation of health and longevity
[6] [2020] The Mechanobiology of Aging
[8] [2019] Personal Communications, Harold Katcher
[9] [2020] Can Blood-Circulating Factors Unveil and Delay Your Biological Aging?
[11] [2013] Studies that Shed New Light on Aging
[12] [2005] Rejuvenation of aged progenitor cells by exposure to a young systemic environment
[13] [2021] Universal DNA methylation age across mammalian tissues
Additional Relevant Articles:
[2021] Models for Evaluation of Biological Age Based on Multiomics
[2021] Epigenetics of Aging and Aging-Associated Diseases
[2021] Attenuation of age-elevated blood factors by repositioning plasmapheresis: A novel perspective and approach
[2021 Common genetic associations between age-related diseases
[2020] Cell non-autonomous regulation of health and longevity
[2020] Correlation analyses between age and indices in routine blood laboratory tests suggest potential aging biomarkers
[2020] Is Oxytocin “Nature’s Medicine
[2020] Data mining of human plasma proteins generates a multitude of highly predictive aging clocks that reflect different aspects of aging
[2020] Personal Communications, Harold Katcher
[2020] Plasma dilution improves cognition and attenuates neuroinflammation in old mice
[2020] Rejuvenation of three germ layers tissues by exchanging old blood plasma with saline-albumin
[2019] Rejuvenation of brain, liver and muscle by simultaneous pharmacological modulation of two signaling determinants, that change in opposite directions with age
[2015] Systemic attenuation of the TGF-β pathway by a single drug simultaneously rejuvenates hippocampal neurogenesis and myogenesis in the same old mammal
[2014] Rumsfeld's Knowns and Unknowns: The Intellectual History of a Quip
[16]. Revising Age: Dual Species measurement of epigenetic age with a single clock; Harold Katcher, Steve Horvath
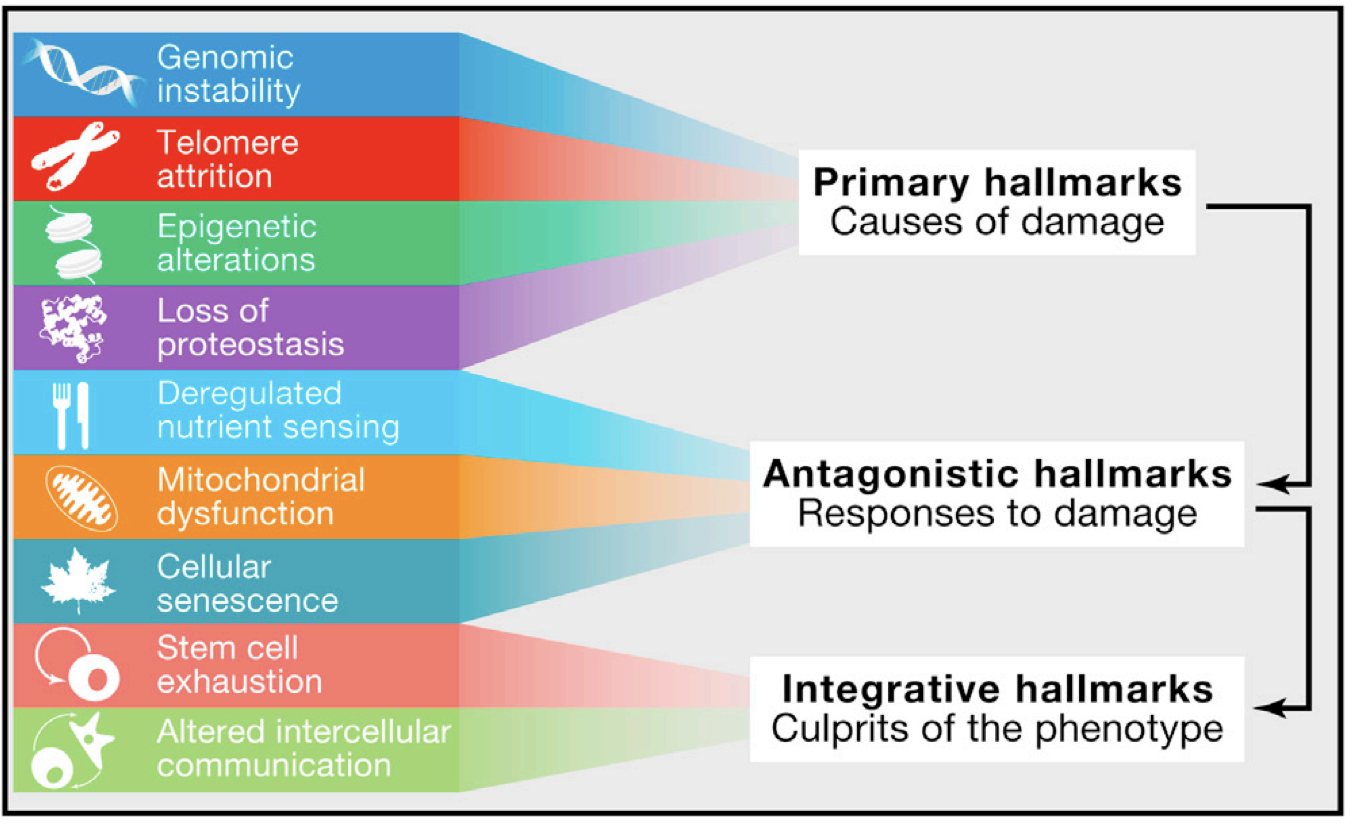
Aging is a complex process that is influenced by a large number of different factors, including genetic, environmental, and lifestyle factors. Here is a partial list of the distinct components of aging, along with references for each one:
◉ Epigenetic changes: Changes in gene expression and regulation that occur as a result of modifications to the DNA molecule or to the proteins with which it interacts, rather than changes to the underlying DNA sequence itself. These changes can affect a wide range of biological processes and contribute to aging and age-related diseases. References:
Fraga, M. F., Ballestar, E., Paz, M. F., Ropero, S., Setien, F., Ballestar, M. L., ... & Esteller, M. (2005). Epigenetic differences arise during the lifetime of monozygotic twins. Proceedings of the National Academy of Sciences, 102(30), 10604-10609.
Zhang, Y., & Chen, R. (2015). Epigenetic modifications and aging. Ageing Research Reviews, 22, 1-12.
◉ Cellular aging: This refers to the changes that occur within cells as they age, including damage to DNA and cellular structures, and the decline in cellular function. References:
de Grey, A. D. N. J. (2007). A roadmap to end aging. Rejuvenation Research, 10(3), 243-255.
Harman, D. (1956). Aging: A theory based on free radical and radiation chemistry. Journal of Gerontology, 11(3), 298-300.
◉ Telomere shortening: Telomeres are the protective end caps on DNA strands that shorten as cells divide. When telomeres become too short, cells can no longer divide and may become damaged, leading to aging and age-related diseases. References:
Rudolph, K. L., Chang, S., Lee, H. W., Blasco, M. A., & Gottlieb, G. J. (1999). Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell, 96(5), 701-712.
Blackburn, E. H., & Greider, C. W. (1985). Identification of a specific telomere terminal transferase activity in tetrahymena extracts. Cell, 43(2), 405-413.
◉ Oxidative stress: This refers to an imbalance between the production of reactive oxygen species (ROS) and the ability of cells to counteract their harmful effects. Oxidative stress can lead to cellular damage and contribute to aging and age-related diseases. References:
Halliwell, B., & Gutteridge, J. M. (1989). Free radicals in biology and medicine. Oxford University Press.
Sies, H. (1997). Oxidative stress: oxidants and antioxidants. Experimental Physiology, 82(2), 291-295.
◉ Inflammation: Chronic inflammation has been linked to aging and age-related diseases, and may contribute to the decline of various physiological systems. References:
Franceschi, C., & Campisi, J. (2014). Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. The Journal of Gerontology: Series A, 69(Suppl 1), S4-S9.
Hotamisligil, G. S. (2006). Inflammation and metabolic disorders. Nature, 444(7121), 860-867.
◉ Nutrient sensing and metabolic changes: Changes in nutrient sensing pathways and metabolism have been linked to aging and age-related diseases. References:
Kaeberlein, M., McDonagh, T., Heltweg, B., Hixon, J., Westman, E. A., Caldwell, S. D., ... & Kennedy, B. K. (1999). Regulation of yeast chronological lifespan by TOR and Sch9 in response to nutrients. Nature, 425(6960), 862-867.
Fontana, L., Partridge, L., & Longo, V. D. (2010). Extending healthy life span—from yeast to humans. Science, 328(5976), 321-326.
◉ Hormonal changes: Changes in hormone levels and the decline in hormone-secreting glands, such as the adrenal glands, thyroid gland, and gonads, have been linked to aging and age-related diseases. References:
Rivas, C., & García-Santos, D. (2015). Role of hormones in aging. In Handbook of Clinical Neurology (Vol. 130, pp. 45-67). Elsevier.
Kelleher, S., Simons, A. D., & Hayes, M. J. (2007). Adrenal insufficiency in the elderly. Age and Ageing, 36(1), 11-16.
◉ Neurodegeneration: The decline in neuronal function and the loss of neurons are key components of aging and are associated with age-related neurodegenerative diseases such as Alzheimer's and Parkinson's disease. References:
De La Torre, J. C. (2002). Alzheimer disease as a neurodegenerative disorder of protein conformation. Annals of the New York Academy of Sciences, 977(1), 74-82.
McKeith, I. G., Galasko, D., Kosaka, K., Perry, E. K., Dickson, D. W., Hansen, L. A., ... & Jellinger, K. (1996). Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): Report of the consortium on DLB international workshop. Neurology, 47(5), 1113-1124.
◉ Skeletal changes: Changes in bone density and structure, along with declines in muscle mass and function, are a key component of aging and can lead to osteoporosis, frailty, and increased risk of falls and fractures. References:
Roux, C., & Kolta, S. (2010). Ageing and the skeleton. European Journal of Endocrinology, 162(2), 201-212.
Buford, T. W., Willoughby, D. S., & Stout, J. R. (2007). The aging of skeletal muscle: the effects of physical activity. Ageing Research Reviews, 6(2), 180-194.
◉ Cardiovascular changes: The decline in cardiovascular function with age, including changes in blood pressure, heart rate, and blood vessel function, can increase the risk of cardiovascular disease. References:
Lakatta, E. G., & Levy, D. (2003). Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part I: aging arteries: a “set up” for vascular disease. Circulation, 107(1), 139-146.
Chaturvedi, N., & Andrews, R. C. (2015). Cardiac ageing. British Medical Bulletin, 115(1), 87-101.
◉ Skin changes: Changes in the skin with age, including wrinkles, sagging, and the loss of skin elasticity, are a visible sign of aging. References:
Kaya, G., & Kocaoğlu, B. (2010). The aging skin. Journal of the Turkish-German Gynecological Association, 11(1), 1-5.
Varani, J., Warner, R. L., & Gharaee-Kermani, M. (2006). Vitamin A and skin aging. Dermatology, 212(2), 176-184.
◉ Cognitive changes: Declines in cognitive function, including memory and processing speed, are a key component of aging and are associated with age-related cognitive decline and neurodegenerative diseases such as Alzheimer's disease. References:
Salthouse, T. A. (2010). When does age-related cognitive decline begin? Neurobiology of Aging, 31(12), 2042-2052.
Ritchie, K., & Kildea, D. (2015). Ageing and cognition. In International Handbook of Ageing and Physical Activity (pp. 203-219). Springer.
◉ Immune system changes: Changes in the immune system with age, including a decline in the number and function of immune cells, can increase the risk of infections and make it more difficult to fight off diseases. References:
Pawelec, G., Derhovanessian, E., & Larbi, A. (2013). The immunological theory of aging. Ageing Research Reviews, 12(1), 627-638.
Fulop, T., Larbi, A., Funato, K., & Pawelec, G. (2015). The role of inflammation in age-related diseases. Ageing Research Reviews, 22, 1-12.
◉ Metabolic changes: Changes in metabolism with age, including changes in insulin sensitivity and glucose metabolism, can increase the risk of age-related diseases such as diabetes and cardiovascular disease. References:
de Groot, L. C. P. G. M., Beck-Peccoz, P., Chrousos, G., Dungan, K., Grossman, A., Hershman, J. M., ... & Lamberts, S. W. J. (2004). Endocrine and metabolic changes in aging. Endocrine Reviews, 25(3), 617-632.
Baur, J. A., Pearson, K. J., Price, N. L., Jamieson, H. A., Lerin, C., Kalra, A., ... & de Cabo, R. (2006). Resveratrol improves health and survival of mice on a high-calorie diet. Nature, 444(7117), 337-342.
◉ Sleep changes: Changes in sleep patterns with age, including difficulties falling asleep and staying asleep, can have negative impacts on health and well-being. References:
Ohayon, M. M. (2002). Epidemiology of insomnia: what we know and what we still need to learn. Sleep Medicine Reviews, 6(2), 97-111.
Ancoli-Israel, S. (2003). The role of sleep in promoting healthy aging and quality of life in older adults. Annual Review of Nursing Research, 21, 287-310.
◉ Gut Microbiome / Probiotics: Microbiome changes: Changes in the composition and diversity of microorganisms in the gut and other parts of the body can contribute to aging and age-related diseases. The microbiome, or the community of microorganisms that live in and on the body, can play an important role in health and disease, and changes in the microbiome with age may impact various physiological processes. References:
Turnbaugh, P. J., Hamady, M., Yatsunenko, T., Cantarel, B. L., Duncan, A., Ley, R. E., ... & Gordon, J. I. (2009). A core gut microbiome in obese and lean twins. Nature, 457(7228), 480-484.
O'Toole, P. W., & Jeffery, I. B. (2016). The microbiome and aging. Microbiology and Molecular Biology Reviews, 80(2), 171-186.
◉ Psychological changes: Changes in psychological well-being with age, including changes in mood, cognition, and personality, can have a significant impact on quality of life. References:
Charles, S. T. (2010). Psychological well-being in adulthood and old age. Annual Review of Psychology, 61, 383-409.
Carstensen, L. L. (2006). The influence of a sense of time on human development. Science, 312(5782), 1913-1915.
◉ Proteomic changes: Changes in the proteins present in cells and tissues, including changes in the levels, types, and functions of proteins, can contribute to aging and age-related diseases. Proteomics is the study of the proteome, or the entire set of proteins present in a cell, tissue, or organism, and can provide insight into the changes that occur during aging. References:
Kurella, V. B., & Iyer, R. (2015). Proteomics in aging research. Ageing Research Reviews, 21, 85-92.
Li, Y., Chen, J., & Liu, X. (2013). Proteomics approaches to aging research. BioMed Research International, 2013, 1-11.
Next page in chronology:
A discussion of the connections between aging and disease