Albumin

(WikiLink: Albumin) - (NLM HUB: Albumin) - (Last Revision: 02/10/22)


Click [√] to Enlarge
Serum albumin levels decreased with age in both men and women. In the cross-sectional study of community-dwelling, self-supported elderly persons. The median value declined from 4.3 g/dL in males aged 65-69 y to 3.9 g/dL A fall in serum albumin concentration was associated significantly with aging. [1] Increases in age related alterations to the remaining albumin are highly correlated with aging.[9]

◉ The primary age regressive benefits from parabiosis experiments, is in all probability “Young and Undamaged,” Albumin.
◉ Dramatic benefits have been documented administering “young” albumin to aged mice and rats. [6]
◉ Recombinant human albumin has been demonstrated to be a safe alternative to plasma derived sources. [6]
◉ One protein subunit of human albumin has almost identical sequence homology to the same biological structure as the copper tripeptide, GHK-Cu.
◉ ⫸It is believed that aging is at least partially caused by the continuous accumulation of damages or unnecessary modifications of proteins [4–6], including free thiol oxidation, carbonylation, advanced glycation end-product (AGE) formation, and homocysteinylation [7–10].
◉ Human serum albumin is the most abundant protein in blood plasma with a serum half-life of about 21 days.[6]
◉ Damages or unnecessary modifications of HSA are related to many pathological conditions and increase with age.
◉ Firstly, the single free thiol in Cys-34 residue of HSA has been proposed to account for approximately 80% of the total free thiols in plasma [12,13], whose oxidation is intimately linked with aging and age-related diseases [14–16].
◉ Secondly, in oxidative environments, carbonyls are also formed especially on the side chains of Pro, Arg, Lys and Thr residues in proteins [17,18].
◉ Elevated carbonyl levels in HSA have been found to be related to aging and varieties of diseases [19–21].
◉ Thirdly, the AGE accumulation of HSA is another important factor found to be involved in aging [9,22]. It is widely reported that AGE formation impairs normal functions of albumin and can induce inflammatory responses, which is connected with aging and the progression of serious diseases [22,23].
◉ Fourthly, it has been widely reported that homocysteine (Hcy) increases with age and is associated with age- related degenerative disorders [10,24–26]. HSA is a major target for homocysteinylation, thus it can efficiently protect other proteins from the toxicity of Hcy [27–29].
Taken together, young and undamaged exogenous rMSA provides a powerful protective function against oxidation of free thiol, carbonylation, AGE formation, and homocysteinylation.⫷[6]
◉ Alkahest Inc., a division of Griffons appears to be perusing this age regressive and disease ammerolating opportunity, with an initial focus on cognitive disorders and frailty (frailty = aging). Frailty being a substitute for reducing the appearance in a search que for the key word of aging. Alkahest specifically indicates in their patent; “BLOOD PLASMA FRACTIONS AS A TREATMENT FOR AGING - ASSOCIATED COGNITIVE DISORDERS” that simply increasing the total amount of albumin is not an ammerolative factor.

Human Serum Albumin is the most abundant protein in the plasma. It now appears that human serum albumin produced in a recombinant bacterial expression systems, [7,8] can deliver pharmacological levels of “young,’ undamaged, reduced or un-oxidized [9] and appropriately folded recombinant Human Serum Albumin (rHSA). When this rHSA is administered to a mammal it results in profound age regressive benefits that include increases in physical strength, mental capacity and increased longevity. These rHSA proteins are devoid of, or incorporate dramatically reduced amounts of age associated, accumulated / unnecessary modifications. These can include; free thiols, carbonation, glycation end-products or homocysteinylation. The resulting recombinant biologically active protein becomes a dramatic age regressive intervention. The expression of these proteins in yeast cultures provides for a relatively inexpensive and limitless supply of these proteins. [10] These humans proteins can also be expressed in plants and lower mammalian species.
▶︎ Here, we report that the injection of exogenous recombinant mouse serum albumin (rMSA) reduced the total damages of serum albumin in C57BL/6N mice, with higher level of free-thiols, lower levels of carbonyls and advanced glycation end-products as well as homocysteines in rMSA-treated mice. The healthspan and lifespan of C57BL/6N mice were significantly improved by rMSA. The grip strength of rMSA-treated female and male mice increased by 29.6% and 17.4%, respectively. Meanwhile, the percentage of successful escape increased 23.0% in rMSA-treated male mice using the Barnes Maze test. Moreover, the median lifespan extensions were 17.6% for female and 20.3% for male, respectively. The rMSA used in this study is young and almost undamaged. We define the concept “young and undamaged” to any protein without any unnecessary modifications by four parameters: intact free thiol (if any), no carbonylation, no advanced glycation end-product, and no homocysteinylation. Here, “young and undamaged” exogenous rMSA used in the present study is much younger and less damaged than the endogenous serum albumin purified from young mice at 1.5 months of age. We predict that undamaged proteins altogether can further improve the healthspan and lifespan of mice. ◀︎[6]
▶︎ Serum free thiols and protein thiols are converted to disulfides with aging [83]. Specifically, Era et al. reported the comparison of serum ALB redox state in young and old subjects (18–24 vs. 60–90 years of age) using HPLC as early as in 1995 [17]. It was found that HMA ratio was obviously higher in the old subjects compared with young subjects. Oettl et al. also presented in their review article that both HNA-1 and HNA-2 ratio increased with aging (ranging approx. 10–100 years of age) [10]. Similarly, Rossi et al. reported increases in cysteinylation and homocysteinylation of plasma proteins with aging in healthy subjects aged 20–93 [84]. Increases in HNA ratios with aging have been further confirmed in recent studies on patients and healthy controls [32,77]. The rationale for these observations has been attributed to oxidative stress accelerated by aging.◀︎[2]
In 2014, Wyss-Coray group reported that plasma from young mice can improve the learning and memory of old mice. Since albumin occupies about 50% of total plasma proteins, it most likely plays the most important role in this process, which was exactly what we found here. In order to achieve the maximal effect of rMSA on longevity, a variety of measures including optimal dosage, frequency, and drug delivery methods are being investigated. We predict that the concept of young and undamaged albumin increasing the longevity can also be applied to any other proteins such as immunoglobulins, fibrinogen, transferrin, transthyretin, and haptoglobin which are major plasma proteins. If so, the combination of young and undamaged major plasma proteins can further increase the longevity. Ideally, all of the young and undamaged plasma proteins altogether can increase the longevity to the largest extent.[6]

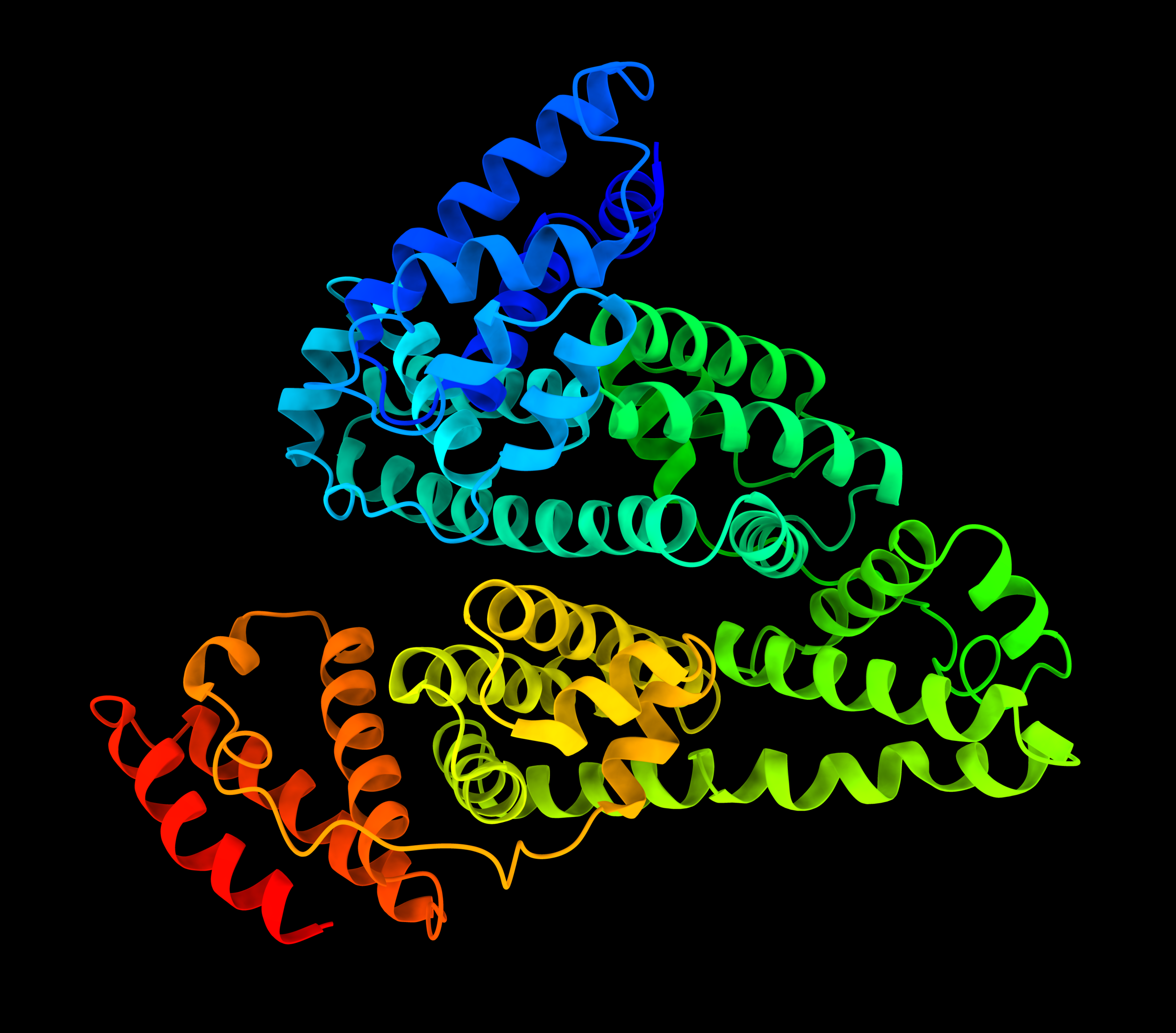
Human Serum Albumin
Human Serum Albumin, Source: Wikipedia Click [√] to Enlarge
Human serum albumin with both Cu(II) and Zn(II) bound in their high-affinity binding sites [4] Click [√] to Enlarge
Serum albumin is able to bind almost all known drugs, nutraceuticals and toxic substances. The protein largely determines their pharmaco- and toxicokinetics, transporting them to target tissues or sites of their biotransformation. Simultaneously, the albumin molecule can bind up to ten ligand molecules, and for this reason, many substances compete with each other for binding sites. In addition to direct competition, the protein is susceptible to allosteric modulation: binding of a ligand in one site can affect the efficiency of binding in another. Due to the thiol group within Cys34, albumin can serve as a trap for reactive oxygen and nitrogen species and participate in redox processes in the body. Moreover, this protein has a number of enzymatic activities: (pseudo) esterase, paraoxonase, phosphotriesterase, thioesterase, glutathione peroxidase and some others. The environment of the bloodstream can significantly affect the functional properties of albumin; the major fatty acids and molecular crowding display the most pronounced influence. This effect should be taken into account when studying the interaction of pharmaceuticals and xenobiotics with albumin in, in vitro and in silico experiments. Interaction with EGL largely determines the integrative role of both albumin and endothelium. All these features suggestthat a targeted influence on albumin to modulate its binding, enzymatic and antioxidant properties can become a kind of adjuvant therapy for pathological conditions. [3]
Serum albumin is the most abundant circulating protein in mammals including humans. It has three isoforms according to the redox state of the free cysteine residue at position 34, named as mercaptalbumin (reduced albumin), non-mercaptalbumin-1 and -2 (oxidized albumin), respectively. The serum albumin redox state has long been viewed as a biomarker of systemic oxidative stress, as the redox state shifts to a more oxidized state in response to the severity of the pathological condition in various diseases such as liver diseases and renal failures. However, recent ex vivo studies revealed oxidized albumin per se could aggravate the pathological conditions. Furthermore, the possibility of the serum albumin redox state as a sensitive protein nutrition biomarker has also been demonstrated in a series of animal studies. A paradigm shift is thus ongoing in the research field of the serum albumin. This article provides an updated overview of analytical techniques for serum albumin redox state and its association with human health, focusing on recent findings. [12]

Therapeutic plasma exchange is the only available way to optimize the plasma albumin of an individual at this time. Please see the TPE page for additional information. Other options are in pre-clinical development. The most exciting Albumin based therapy is described in “Young and Undamaged rMSA Improves the Healthspan and Lifespan of Mice”


🔷 ? 🔷

🔷 ? 🔷
🔷 ? 🔷


🔷 ? 🔷

🔷 ? 🔷

[2] [2019] Biochemistry and Cell Biology of Ageing: Part II
[4] [2018] A Molecular Depot: Human Serum Albumin Carries Essential Cu and Zn in Serum
[6] [2021] Young and Undamaged rMSA Improves the Healthspan and Lifespan of Mice
[7] [2005] Summary of recombinant human serum albumin development
[9] [2021] Serum Albumin Redox States: More Than Oxidative Stress Biomarker
[12] [2021] Serum Albumin Redox States: More Than Oxidative Stress Biomarker
[13] [2018] A Molecular Depot: Human Serum Albumin Carries Essential Cu and Zn in Serum
[19] [2013] The mechanisms of action of plasma exchange
[20] [2019] Biochemistry and Cell Biology of Ageing: Part II
[21] [2020] Reversing age: dual species measurement of epigenetic age with a single clock
◉ PATENTS
[P1] [2020] USPTO US 10,525,107B2; Alkerest; BLOOD PLASMA FRACTIONS AS A TREATMENT FOR AGING - ASSOCIATED COGNITIVE DISORDERS
[P2] [2022] Int Patent #: 1.WO2022150818: Yuven Research;ANTI-AGING COMPOSITIONS AND USES THEREOF

◉ REFERENCES